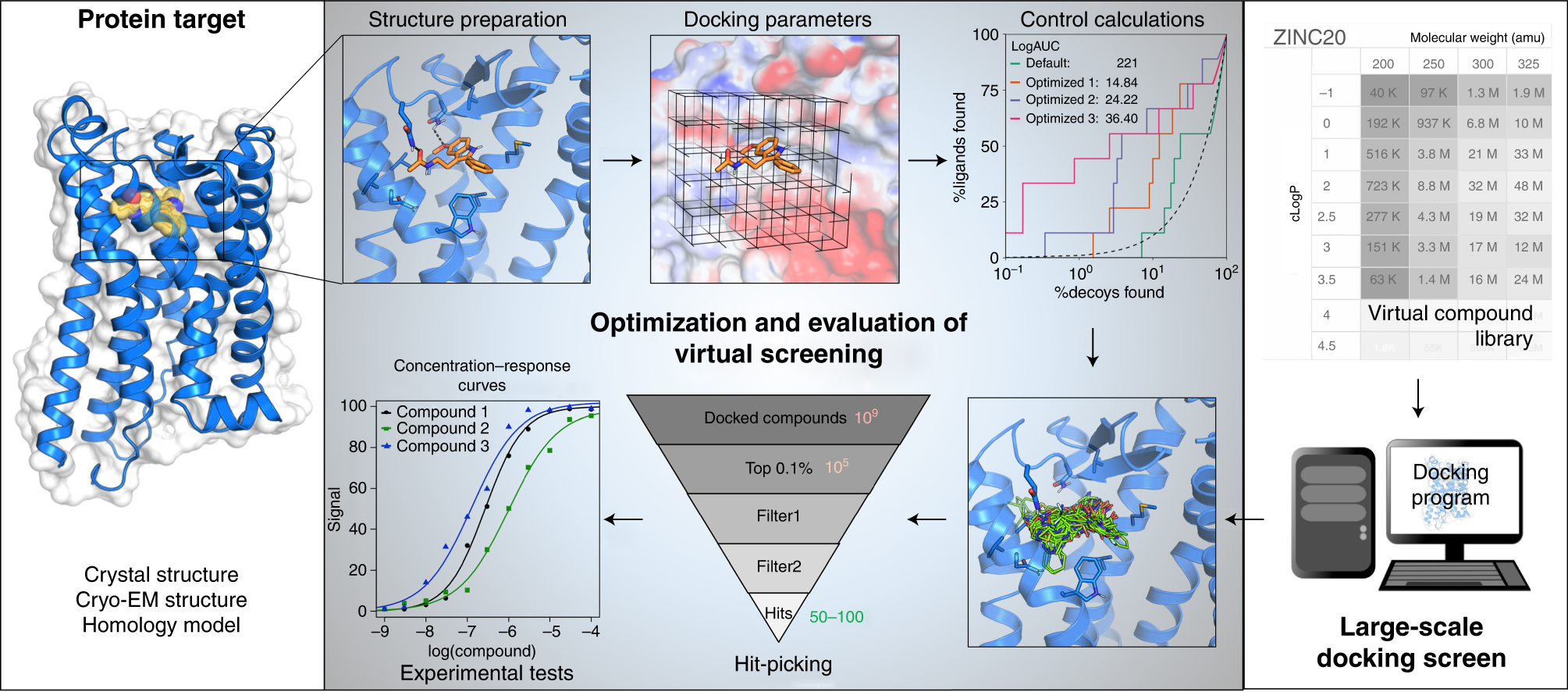
Molecules | Free Full-Text | Ensemble Docking Coupled to Linear Interaction Energy Calculations for Identification of Coronavirus Main Protease (3CLpro) Non-Covalent Small-Molecule Inhibitors | HTML
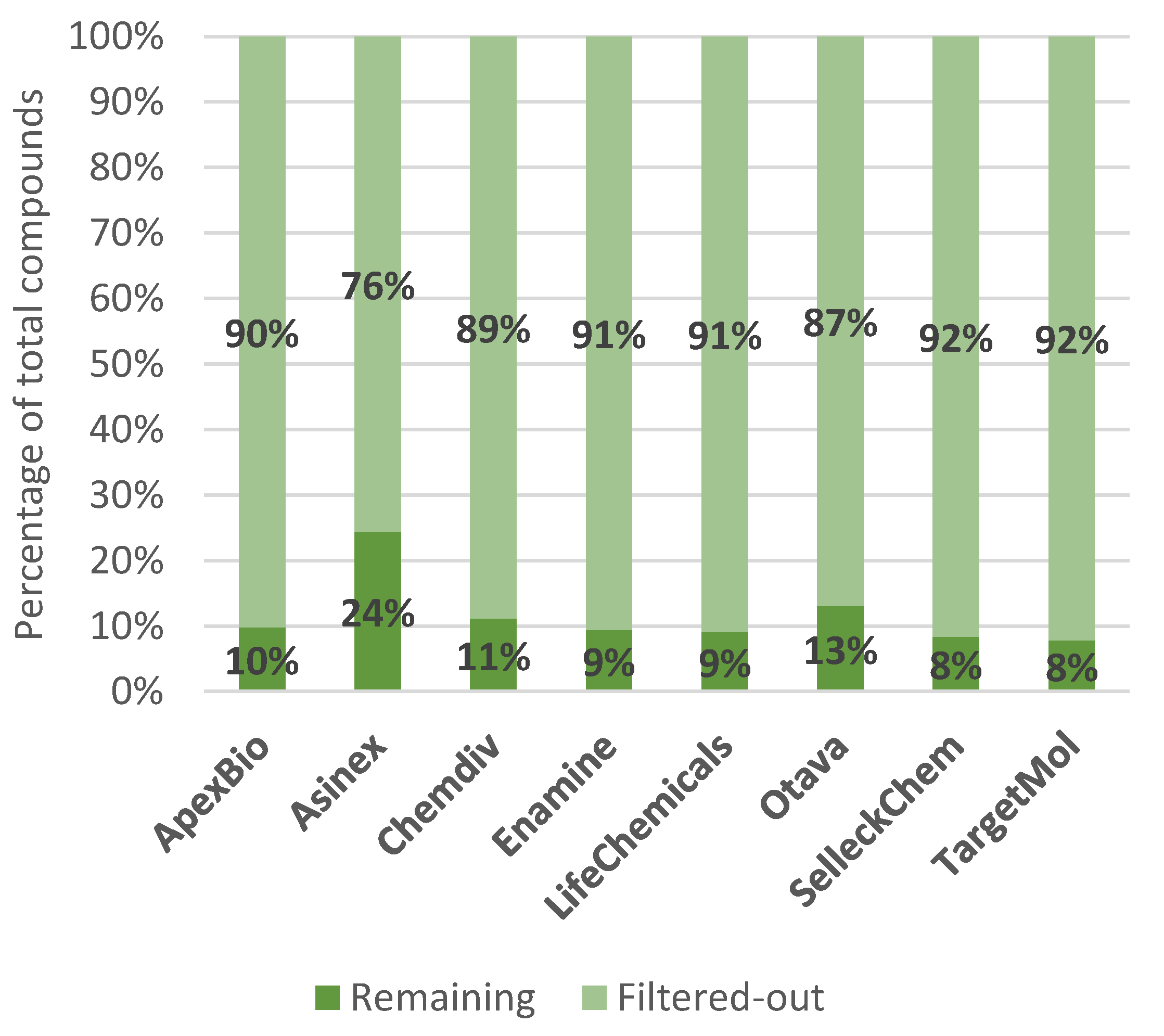
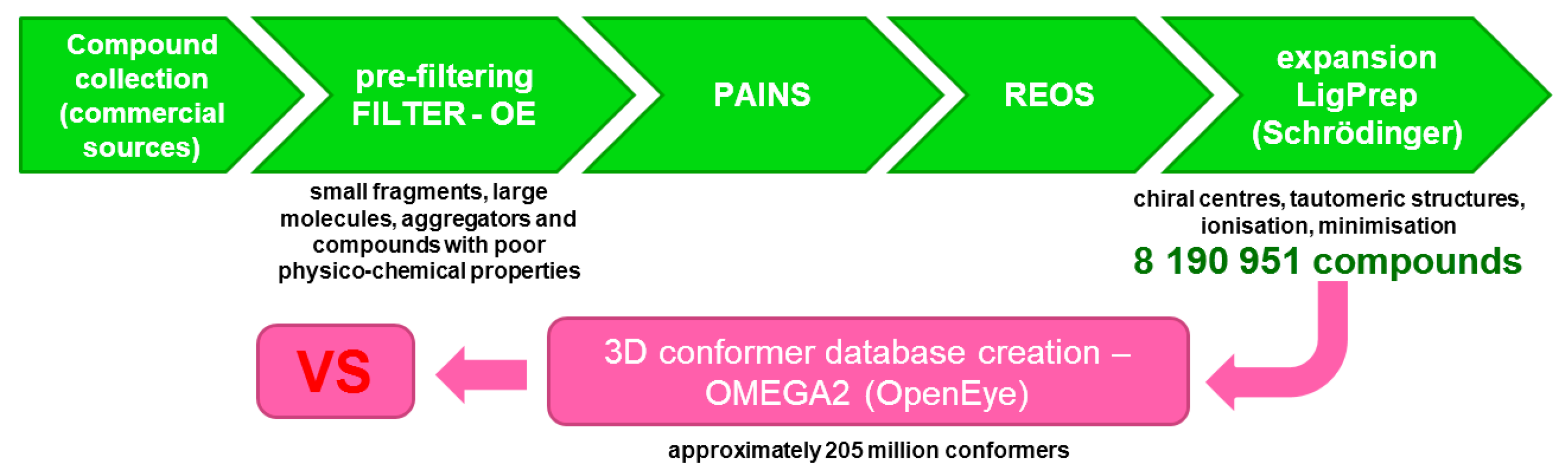
IJMS | Free Full-Text | Commercial SARS-CoV-2 Targeted, Protease Inhibitor Focused and Protein–Protein Interaction Inhibitor Focused Molecular Libraries for Virtual Screening and Drug Design | HTML

Highly Promiscuous Small Molecules from Biological Screening Assays Include Many Pan-Assay Interference Compounds but Also Candidates for Polypharmacology | Journal of Medicinal Chemistry
Highly Promiscuous Small Molecules from Biological Screening Assays Include Many Pan-Assay Interference Compounds but Also Candi

VSPrep: A General KNIME Workflow for the Preparation of Molecules for Virtual Screening - Gally - 2017 - Molecular Informatics - Wiley Online Library

Molecules | Free Full-Text | Identifying Promiscuous Compounds with Activity against Different Target Classes | HTML

IJMS | Free Full-Text | Potential Novel Thioether-Amide or Guanidine-Linker Class of SARS-CoV-2 Virus RNA-Dependent RNA Polymerase Inhibitors Identified by High-Throughput Virtual Screening Coupled to Free-Energy Calculations | HTML
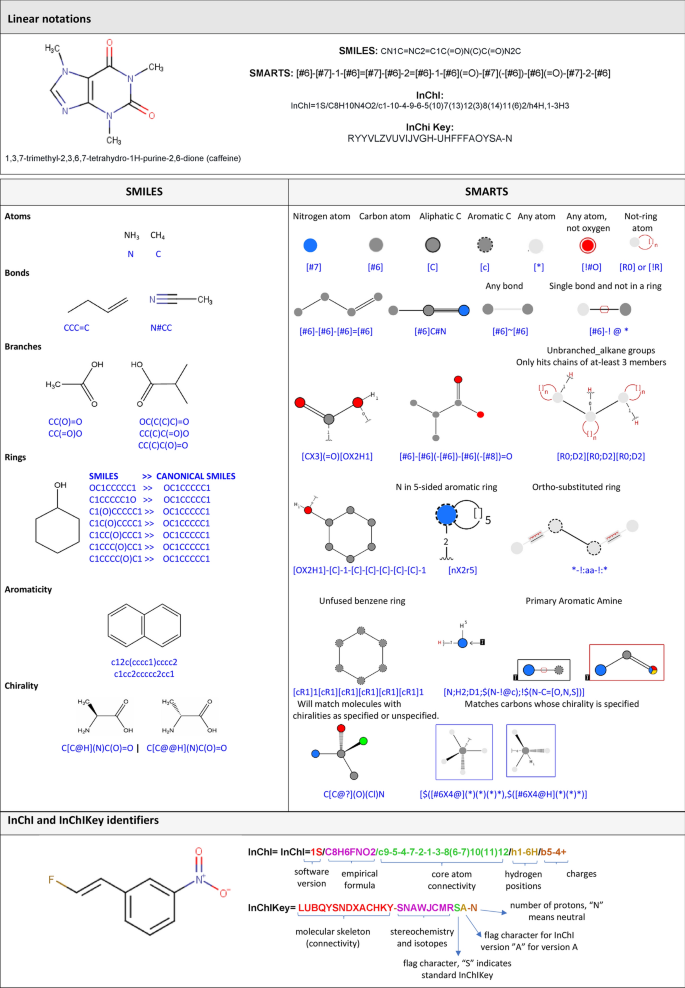
Chemoinformatics-based enumeration of chemical libraries: a tutorial | Journal of Cheminformatics | Full Text

Amazon.com : 6MP PTZ IP Camera with Pan Tilt 3X Optical Zoom,Outdoor Security Camera, Built in Mic,Human/Vehcile Detection, 98ft IR Night Vision,IP66& IK10,Compatible for Hikvision(PTZ-3601-IZ) : Electronics

How Frequently Are Pan-Assay Interference Compounds Active? Large-Scale Analysis of Screening Data Reveals Diverse Activity Profiles, Low Global Hit Frequency, and Many Consistently Inactive Compounds | Journal of Medicinal Chemistry

Highly Promiscuous Small Molecules from Biological Screening Assays Include Many Pan-Assay Interference Compounds but Also Candidates for Polypharmacology | Journal of Medicinal Chemistry
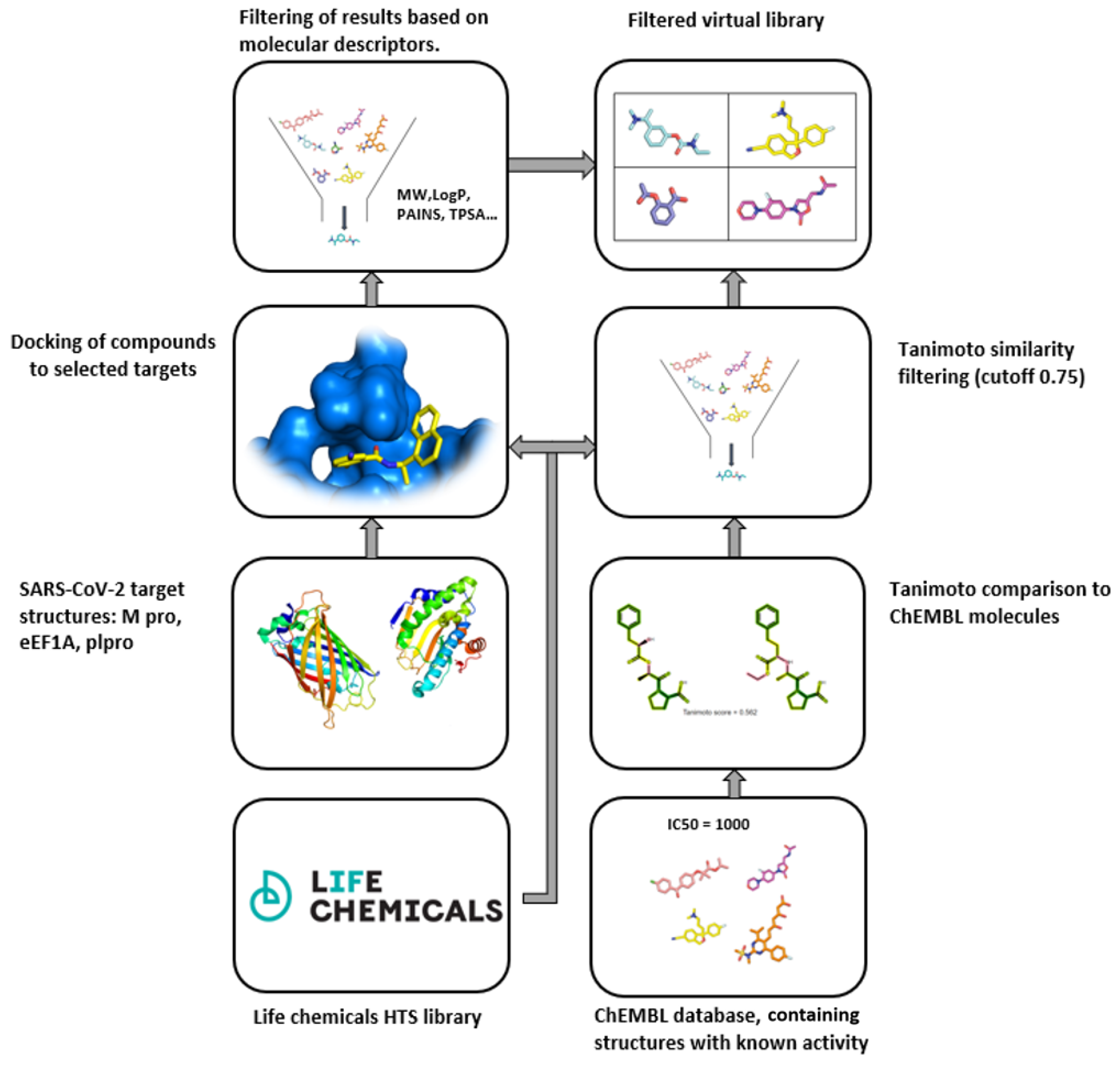
IJMS | Free Full-Text | Commercial SARS-CoV-2 Targeted, Protease Inhibitor Focused and Protein–Protein Interaction Inhibitor Focused Molecular Libraries for Virtual Screening and Drug Design | HTML

Artificial Intelligence Applied to the Rapid Identification of New Antimalarial Candidates with Dual‐Stage Activity - Lima - 2021 - ChemMedChem - Wiley Online Library

Machine learning classification can reduce false positives in structure-based virtual screening | PNAS
Natural bioactive compounds as a new source of promising G protein-coupled estrogen receptor (GPER) modulators: comprehensive in

Structural dynamics and inhibitor searching for Wnt-4 protein using comparative computational studies. - Abstract - Europe PMC
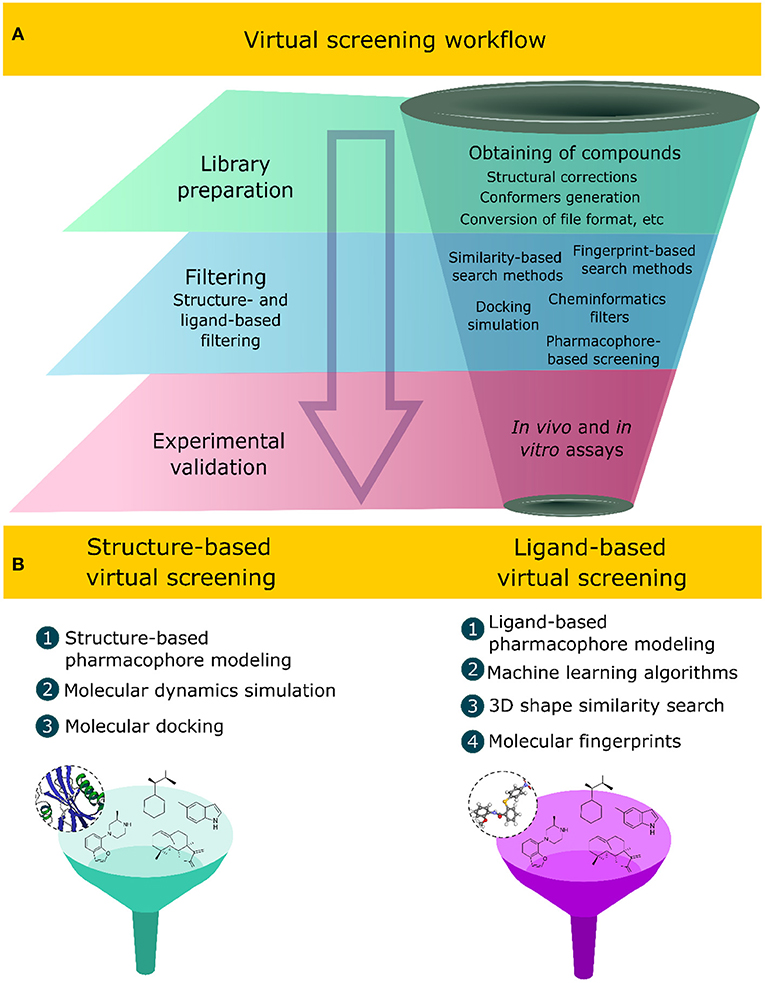
Frontiers | Applications of Virtual Screening in Bioprospecting: Facts, Shifts, and Perspectives to Explore the Chemo-Structural Diversity of Natural Products
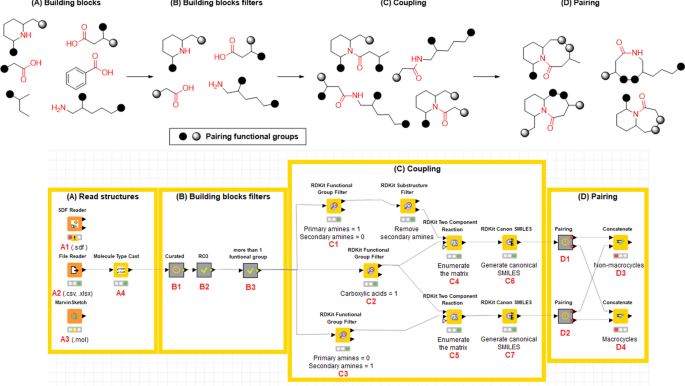
Chemoinformatics-based enumeration of chemical libraries: a tutorial | Journal of Cheminformatics | Full Text
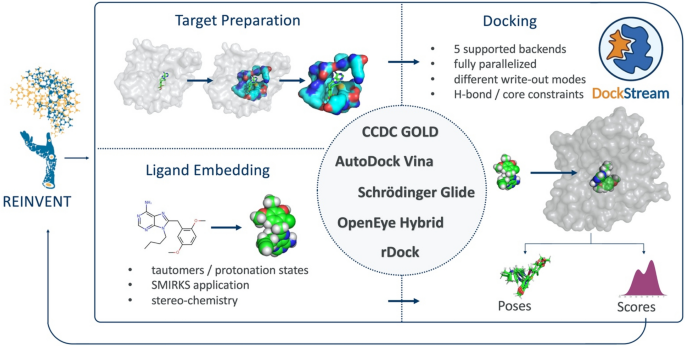
DockStream: a docking wrapper to enhance de novo molecular design | Journal of Cheminformatics | Full Text